Mixture fraction
General properties
- A measure of local stoichiometry
- A measure of mixing
- Very common in nonpremixed flames, especially turbulent flames
- Combustion is between two streams: Fuel and Oxidizer
- Stream 0 is typically oxidizer (air)
- Stream 1 is typically fuel
- The fuel and air compositions are arbitrary. Mixture fraction in generically described in terms of any two streams.
- Consider a turbulent nonpremixed flame
- full range of mixtures between pure air and pure fuel
- Highly complex
- Temperature, composition, velocity, soot, radiation, etc.
- If we can relate properties to a local state of mixing, then we can significantly simplify the representation of the flame
- Assuming products of complete combustion (and uniform species diffusivities), if we know the mixture fraction, we know the full composition. If we assume adiabatic combustion, then if when know the mixture fraction, we also know the temperature.
Simple definition
Take $F$ and $A$ to be masses of fuel and air. Then,
$$\xi = \frac{F}{F+A},$$$$1-\xi = \frac{A}{F+A},$$
the ratio of these gives
$$\frac{F}{A} = \frac{\xi}{1-\xi}.$$Note that:
- $\xi=0$ in the oxidizer stream.
- $\xi=1$ in the fuel stream.
Write in terms of equivalence ratio
$$\phi = \frac{F/A}{(F/A)\_{st}} = \frac{\xi/(1-\xi)}{\xi\_{st}/(1-\xi\_{st})} = \frac{\xi(1-\xi\_{st})}{\xi\_{st}(1-\xi)}.$$Opposed jet flame
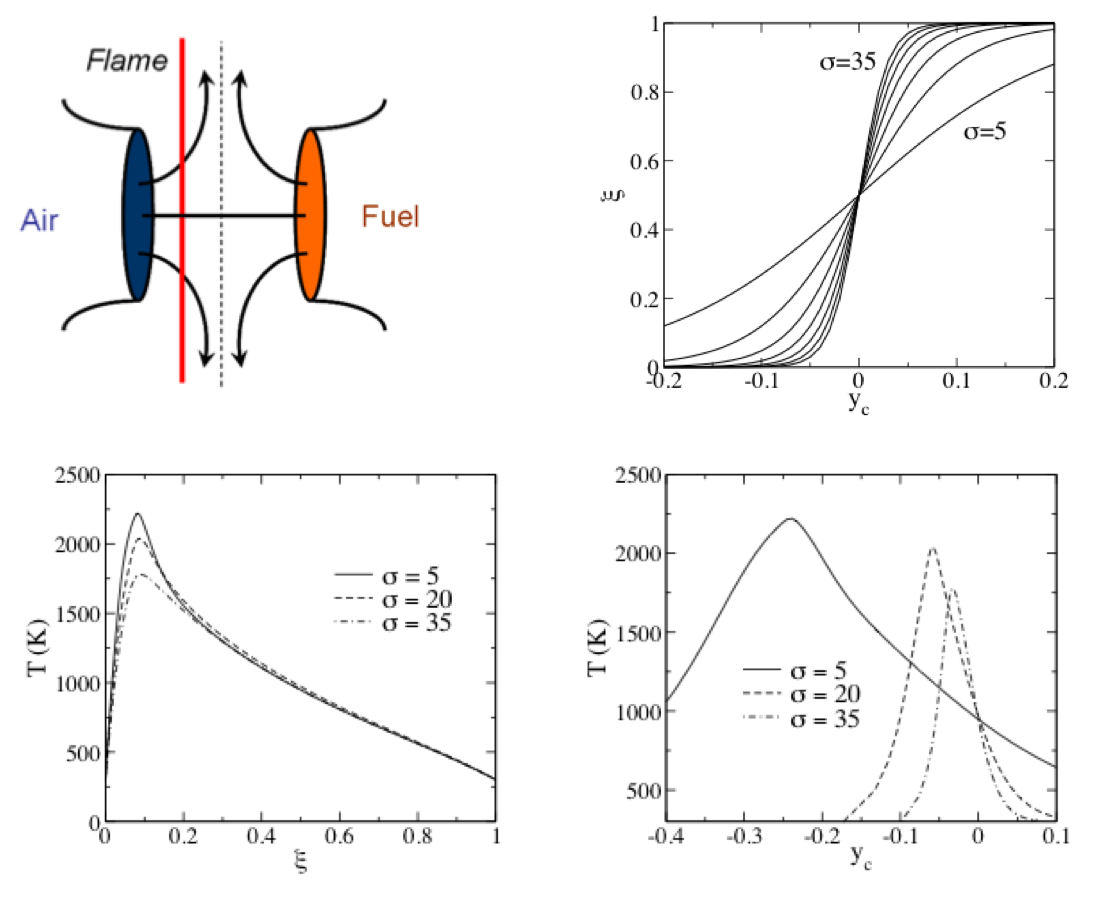
Consider the above opposed jet diffusion flame.
- Note the the flame lies on the oxidizer side of the centerline.
- The flow stagnates at the centerline.
- The top right plot shows the mixture fraction profile versus position for various strain rates, related to the $\sigma$ width parameter, which is related to the jet velocities.
- We can plot the flame temperature versus position (here, $y_c$ is position). See the lower right plot.
- But since there is a one-to-one relation between $\xi$ and $y_c$, we can plot $T$ versus $\xi$.
- This is done in the lower left plot.
- Note how the curves collapse much more strongly for $T(\xi)$ than for $T(y_c)$.
- It is very common in combustion modeling to use a mixture fraction coordinate instead of a spatial coordinate.
Mixture fraction with reaction
The mixture fraction represents the extent of mixing between two streams.
What happens when those two streams react?
- $\xi = F/(F+A)$ but we don’t have F, A, we have F, A, Products.
- $\xi$ is just the state of mixing between the two streams $\xi=0$ and $\xi=1$, and that has not changed.
- So, we should be able to compute it.
- We defined $\xi$ as $F/(F+A)$, but that is not quite right. It is really
$$\xi = \frac{m_1}{m_1+m_0}$$
- That is, $\xi$ is the fraction of local mass that originated in stream $\xi=1$.
- Whether that mass is represented as unburned reactants, or burned (or parially burned) products does not change $\xi$.
Consider $H_2 + \frac{1}{2}O_2 \rightarrow H_2O.$
- If $\xi=0=O_2$ and $\xi=1=H_2$, then
- Look at the atoms: $HH + O \rightarrow HOH.$
- Reaction has not changed the mass fraction originting in the $\xi=1$ or Fuel stream. This is the mass fraction of $H$ here.
Mass is not created or destroyed, so define $\xi$ in terms of conserved quantities: elements
-
Mass fraction of carbon: $y_C$.
-
Denote FS as “fuel stuff” (or mass that originated in the $\xi=1$ stream)
-
Denote AS as “air stuff” (or mass that originated in the $\xi=0$ stream). or,
$$y_C = (\xi)\cdot y_{C,\xi=1} + (1-\xi)\cdot y_{C,\xi=0}$$ -
The mass fraction of carbon is a linear combination of the mass fractions of carbon in the $\xi=0$ (air) stream, and the $\xi=1$ (fuel) stream.
-
solve for $\xi$
$$\xi = \frac{y_C - y_{C,\xi=0}}{y_{C,\xi=1}-y_{C,\xi=0}}$$
These equations hold for any conserved quantity $\beta$, not just mass of carbon.
- Typical conserved quantities are mass of the elements: carbon, hydrogen, nitrogen, oxygen, etc.
- For adiabatic systems, enthalpy is also conserved.
- Also, any linear combination of conserved quantities is also conserved.
$$\xi = \frac{\beta - \beta_{\xi=0}}{\beta_{\xi=1}-\beta_{\xi=0}}$$
- This tells us how to compute $\xi$ for any mixture, whether reacted or not.
- We define the composition of the $\xi=0$ stream (usually air), and the $\xi=1$ stream (usually pure fuel).
Example—methane/air flame
Find the stoichiometric mixture fraction $\xi_{st}$.
$$CH_4 + 2(O_2 + 3.76 N_2)\rightarrow CO_2 + 2H_2O + 7.52 N_2$$- Let $\xi=1=CH_4$, and $\xi=0=\mbox{air}$
- $\xi_{st}$ is the same before and after reaction, so evaluate using reactants since the reactants are directly in terms of the streams and we don’t have any mixing of the fuel and air streams among species like we do for the products.
That is, in a stoichiometric mixture there is about 20 times more air than fuel (mass).
Compute $\xi$ from measured species
Suppose we measure the mass fractions of CO$_2$, H$_2$O, N$_2$, O$_2$, CH$_4$ at a point in a flame. Find the local $\xi$.
- The fuel stuff and the air stuff are mixed among the species (CO$_2$ has carbon from the fuel mixed with oxygen from the air).
- Use the previous relation: $$\xi = \frac{\beta - \beta_{\xi=0}}{\beta_{\xi=1}-\beta_{\xi=0}}$$
- Let $\beta = y_C$
- Need $y_C$, $y_{C,\xi=0}$, and $y_{C,\xi=1}$.
- Carbon appears in species CO$\_2$ and CH$\_4$, so
$$y_C = y_{CO2}\frac{1\cdot M_C}{M_{CO2}} + y_{CH4}\frac{1\cdot M_C}{M_{CH4}}$$
- That is, the mass fraction of carbon is (the mass fraction of CO$_2$ times the mass of carbon per mass of CO$_2$) plus (the mass fraction of CH$_4$ times the mass of carbon per mass of CH$_4$).
- We evaluate this expression at the given point in the flame (using the previous equation), and in the fuel stream, and in the air stream, where the previous equation reduces to.
- In the fuel stream, $y_{C,\xi=1} = M_C/M_{CH4}$.
- In the air stream, $y_{C,\xi=0} = 0$.
We will get the same answer if we use $\beta=y_H$ or $\beta=y_O$, or $\beta=y_N$, or (for adiabatic) $\beta=h,$ or for any linear combination of these.
- Using $\beta = y_N$ is the easiest since the only species that contains N is N$_2$.
- This is not always true. There can be N in the fuel, and other minor species like NO, NO$_2$, etc.
Differential diffusion
Actually, this statement (about getting the same $\xi$ for various $\psi$) is only strictly true when species have the same diffusivities.
- Consider CH$_4$ burning in O$_2$. If species diffusivities are all the same, then we will always have a 4:1 H:C ratio, for any extent of mixing. However, suppose H$_2$ is one of the intermediate species. It has a high diffusivity, and will leave the carbon containing species behind, changing the 4:1 H:C ratio. A mixture fraction computed using $\beta=y_H$ will be different that one computed using $\beta=y_C$.
A direct simulation of a turbulent ethylene jet flame was computing with differential diffusion. 19 species were tracked. At every point in the flame the mass fractions of all species were computed and various mixture fractions computed. These are shown in the parity plot below, where each mixture fraction is plotted versus the carbon mixture fraction.
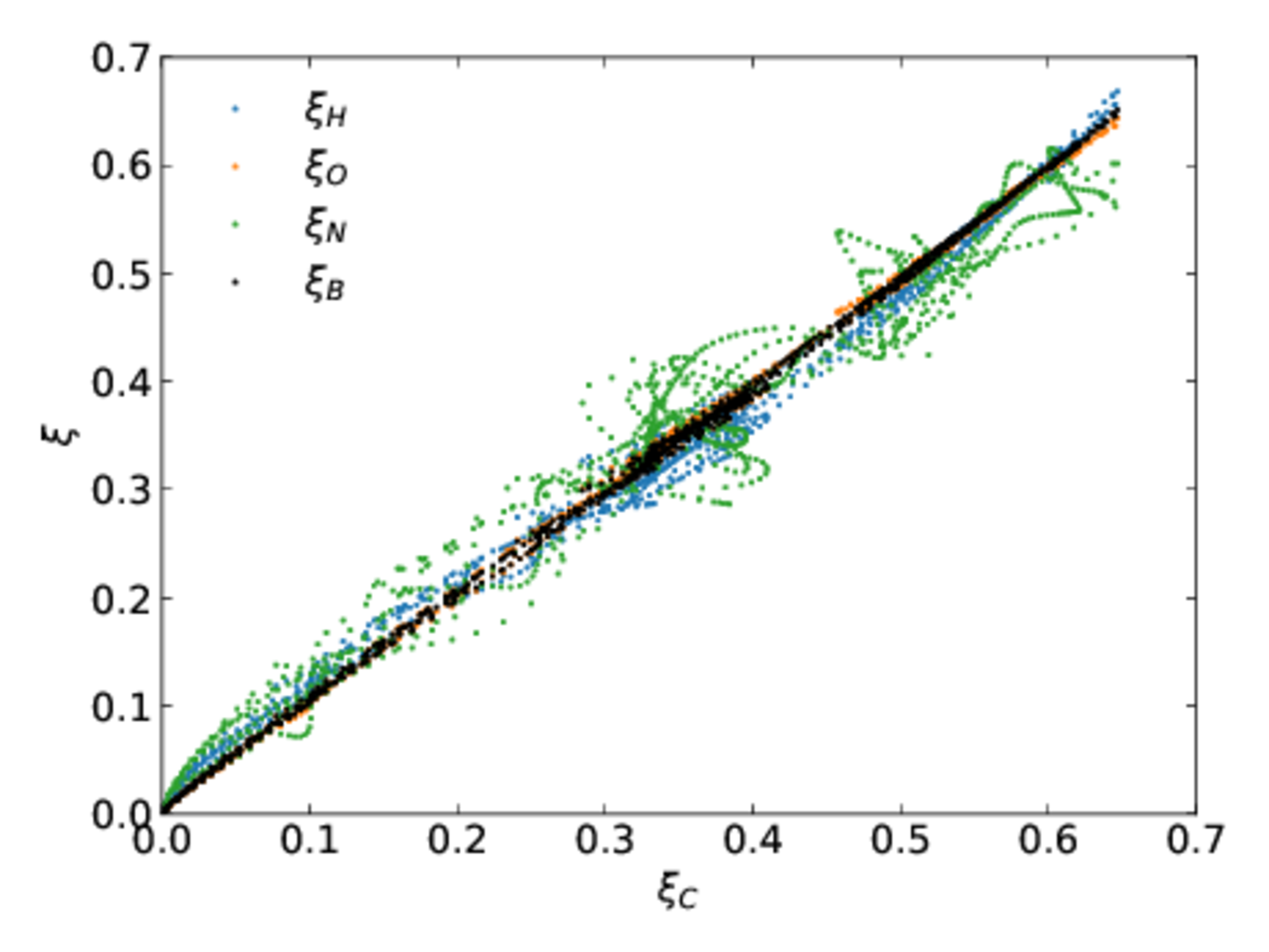
- Without differential diffusion, all points would lie on a 45 degree line.
- $\xi_B$ is the Bilger mixture fraction, which is very popular. $\xi_B$ uses $$\beta = \frac{2}{M_C}y_C + \frac{1}{2M_H}y_H - \frac{1}{M_O}y_O.$$
Note
- For products of complete combustion, species mass fractions are linear with mixture fraction on either side of $\xi_{st}$.
- This means that if you know the species mass fractions at $\xi=0$ and at $\xi=1$ (which you do from the stream definition), and if you know it at $\xi=\xi_{st}$, which is just from the balanced reaction equation, then the composition at every other $\xi$ is easy to find from drawing lines between $y_{k,\xi=0}$ and $y_{k,\xi=\xi_{st}}$, or between $y_{k,\xi=\xi_{st}}$ and $y_{k,\xi=1}$.
